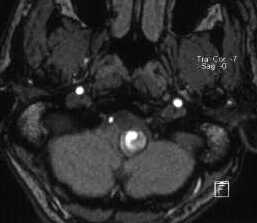
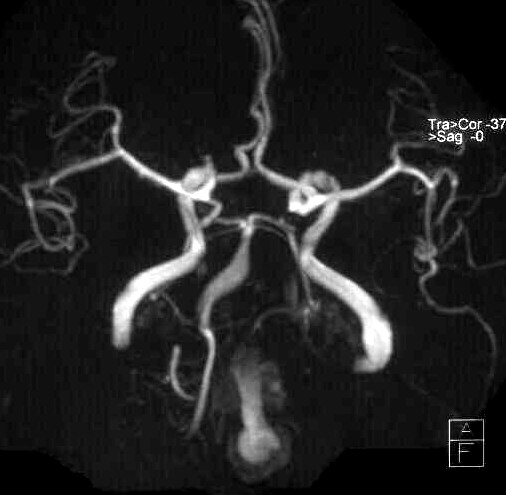
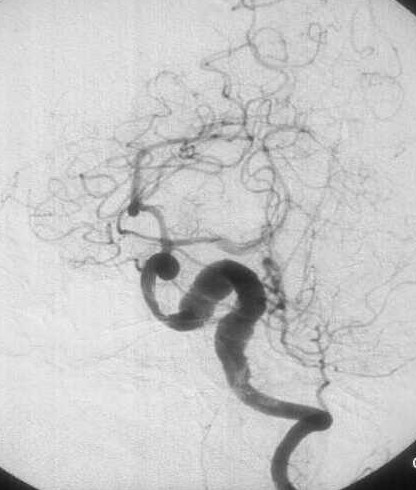
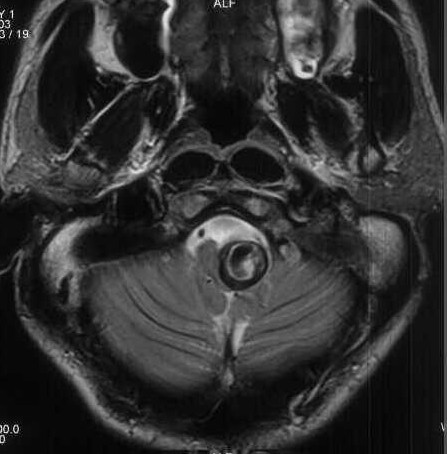
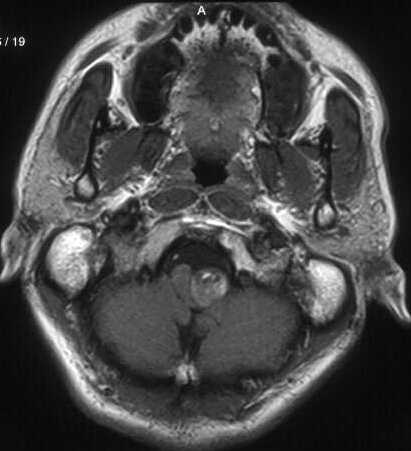
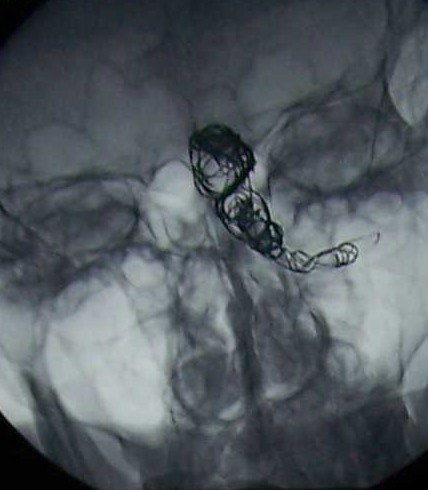
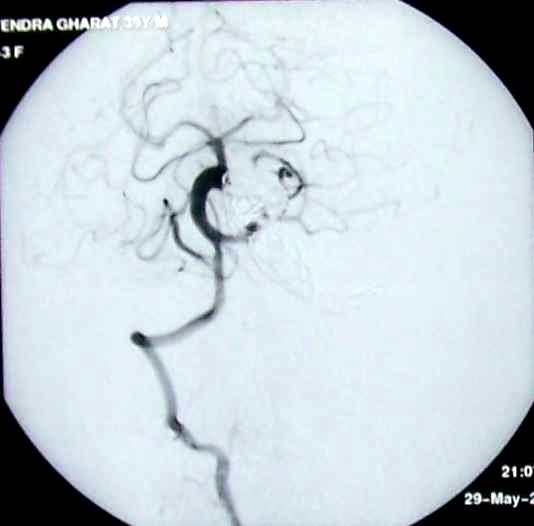
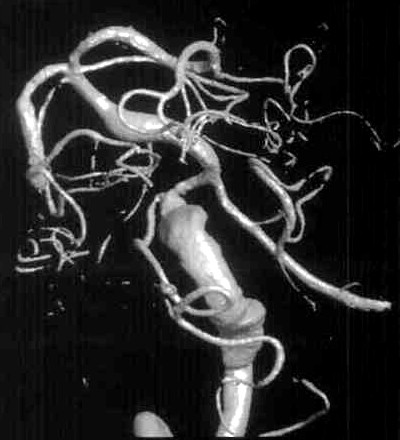
Diagnosis - Giant partially thrombosed dissecting intra dural left VA Aneurysm
Dissecting aneurysms of the intracranial VA are dynamic ; ruptured lesions may manifest with severe SAH and devastating neurological sequelae and have a proclivity to repeated hemorrhage. Similarly, unruptured lesions have a propensity to enlarge and produce symptoms due to both compressive and embolic mechanisms.Approximately 30–70% of patients with rupture will have rebleeding, while smaller percentages will stabilize or improve without treatment.
Symptoms of vertebrobasilar dissecting aneurysms depend on factors such as size and location, including whether the lesion is intradural or extadural. Typical presentations include SAH, thromboembolic events, and cranial neuropathy from mass effect. Wallenberg syndrome (lateral medullary syndrome), Cheiro-oral syndrome, hemifacial spasm, and Foville syndrome (brain stem infarct) can all be related to vertebrobasilar dissecting aneurysms.
Pathophysiology : Histologically, the intradural segment of the vertebral artery has a thin media and adventitia with fewer elastic fibers, so dissections of the intradural vertebral artery are more likely to result in SAH with pseuduoaneurysm compared with dissections of the extradural segment. Also, vasa vasorum are diminished, which may limit healing.
An expanding hematoma in the vessel wall is the root lesion in VAD. This intramural hematoma can arise spontaneously or as a secondary result of minor trauma, through hemorrhage of the vasa vasorum within the media of the vessel. It also can be introduced through an intimal flap that develops at the level of the inner lumen of the vessel.
This intramural hemorrhage can evolve in a variety of ways, resulting in any of the following consequences:
1. The hematoma may seal off and, if sufficiently small, remain largely asymptomatic.
2 If the dissection is subintimal, the expanding hematoma may partially or completely occlude the vertebral artery or one of its branches. Extensive dissections (those that extend intracranially and involve the basilar artery) result in infarctions of the brain stem, cerebellum or, rarely, the spinal cord. Subintimal dissections also may rupture back into the vertebral artery, thus creating a false lumen (pseudolumen).
3. Subadventitial dissections tend to cause pseudoaneurysmal dilation of the vertebral artery, which may compress adjacent neurologic structures. These subadventitial dissections are prone to rupture through the adventitia, resulting in subarachnoid hemorrhage. In an autopsy series of more than 100 patients with subarachnoid hemorrhage, 5% of the hemorrhages were deemed the result of VAD.
4. The intimal disruption and low flow states that arise in VAD create a thrombogenic milieu in which emboli may form and propagate distally. This results in transient ischemia or infarction.
Vertebral artery anatomy: The course of the vertebral artery usually is divided into 4 sections as follows:
Segment I runs from its takeoff at the first branch of the subclavian artery to the transverse foramina of cervical vertebra C5 or C6.
Segment II runs entirely within the transverse foramina of C5/C6 to C2.
Segment III, a tortuous segment, begins at the transverse foramen of C2, runs posterolaterally to loop around the posterior arch of C1, and passes subsequently between the atlas and the occiput. This segment is encased in muscles, nerves, and the atlanto-occipital membrane.
Segment IV, the intracranial segment, begins as it pierces the dura at the foramen magnum and continues until the junction of the pons and medulla, where the vertebral arteries merge to join the larger proximal basilar trunk.
Spontaneous dissection of the vertebral artery usually occurs in the tortuous distal extracranial segment (segment III) but may extend into the intracranial portion or segment IV.
Diagnosis :
Magnetic resonance imaging
1. MRI detects both the intramural thrombus and intimal flap that are characteristic of VAD.
2. Hyperintensity of the vessel wall seen on T1-weighted axial images is considered by some to be pathognomonic of VAD
Cerebral Angiography
These lesions have a variable angiographic appearance, most often demonstrating a region of vessel tapering and then enlargement, also known as a “pearl and string†configuration. This particular configuration has the highest propensity for re rupture. The V4 aneurysms may also demonstrate fusiform dilation, severe vessel narrowing, and even occlusion. Irregularities such as aneurysm dilation and vessel tapering actually facilitate transarterial coil placement, because both features allow the coils to conform to the diseased segment.
Angiograms are assessed for size, shape, and location of the dissecting aneurysm with respect to the major branches and collaterals (ie, the presence or absence of the contralateral vertebral artery or posterior communicating arteries). Each lesion should be examined for evidence of extension of the dissection into adjacent arterial segments, including the PICA and basilar artery. The perforator vessels and the anterior spinal artery should be identified.
Each dissecting aneurysm can be classified as follows:
1. Lesion inferior to the origin of the PICA,
2. Lesion involving the vertebral artery without antegrade filling of the PICA or anterior spinal artery visualized,
3. Lesion of the vertebral artery involving the PICA or anterior spinal artery with patency of this branch,
4. Lesion of the supra-PICA vertebral artery with or without extension into the vertebrobasilar junction, and
5. Lesion of the basilar artery.
Management :
Initially, V4 aneurysms were treated surgically with hunterian ligation of the proximal artery. Now, this is no longer practiced.
Endovascular Management is preferred.
Standard endovascular methods for dissecting intradural VA aneurysms include:
1. Parent artery coil occlusion (proximal occlusion with flow reversal ) ,
2. Aneurysm coil occlusion,
3. Stent placement, and
4. Trans stent coil delivery.
- Proximal occlusion (with flow reversal) of the affected vertebral artery, using coils or balloons, is commonly used. The advantage of this technique is that it limits catheter and wire manipulation across a narrow or irregular segment and may allow for better collateral circulation, especially when lesions involve the origin of the PICA or anterior spinal artery. This technique also uses fewer coils, with a lower cost as a result. It should also be considered for lesions with configurations unlikely to hold coils in a stable position. These include fusiform or wide-necked pseudoaneurysms. Nevertheless, proximal parent artery occlusion is not believed to completely eliminate the risk of rebleeding.
- Flexible intravascular stents followed by coil embolization of the pseudoaneurysm may be used to preserve the lumen especially if the dominant vertebral artery is affected.
Follow-up angiography, gadolinium-enhanced MR angiography, or CT angiography can be performed at 6–12 months to determine whether the affected segment is smaller or healed. ]
In the above case, Proximal occlusion of left VA was performed sparing the PICA using detachable platinum coils.